kilemall
SOC-14 5K
In other words lost in the 20 dt of the Bridge.
I always figured part of the stateroom volume budget.
In other words lost in the 20 dt of the Bridge.
An interesting add here would be a block and tackle system at the top to give normal people a chance of lifting someone up in that harness. Doesn't have to be super elaborate, but you'll need more than just a pulley. In the end it can just be an eyelet mounted to the roof, then the pulley system can be hung in an emergency, stored in a box nearby vs "DOH! Clonked my head on that @&#$^@ pulley thing again!". Who doesn't love lose, swinging masses in a starship.Now I have even more to think about.
Gravity ... OFF.An interesting add here would be a block and tackle system at the top to give normal people a chance of lifting someone up in that harness.
Before or after you crashed on the planet?Gravity ... OFF.
large amounts of mass, when moving even in zero G, are still large amounts of mass. 1 ton machine going 1 meter/second is still going to crush you if you get between it and the wall.Gravity ... OFF.
Don't need to use massive amounts of strength to move large masses in low/zero-g conditions ...
May want something sturdier than a "thin piece of string" ... but you get the idea ...
But that store of oxygen in the ship's spaces is not consumed. It's a one-off requirement, and as long as there's adequate air circulation consumed oxygen can be replaced at a single point (and the CO2, etc. scrubbed out).Way more is needed than that since every crewed volume of the ship has to be able to supply the oxygen, unless the crew wear vacc suits for the whole time in ship.
Most explosives include their own oxidizers. There are exceptions.I wonder if oxygen becomes necessary as an environmental factor, in all these attempts at ingress and egress?
No. You do not use 100% O2 onboard atmosphere.I think that would be pure, undiluted oxygen.
Optionally, if temperature isn't that much of an issue, you can get a four hour duration oxygen bottle from the ship's locker, and put on either a tee shirt, or a pullover.
Actually ... there is a modern counter-example in which 100% O2 was used to solve a variety of potential engineering issues concerning flight.No. You do not use 100% O2 onboard atmosphere.
We learned that the hard way with Apollo 1 in 1967. (Wikipedia)
The rationale behind this choice was that 100% O2 at reduced pressure (PSI) delivered an equivalent quantity of O2 in a lower % fraction at a higher pressure (PSI). It was basically trading O2 fraction for internal suit pressure.Within an hour of launch, the crew began a pre-breathing protocol to reduce nitrogen in their bodies and minimize the risk of decompression sickness during the planned spacewalk on day three. Over three days, the cabin pressure gradually decreased from 14.5 to 8.6 pounds per square inch (100 to 59 kPa) while oxygen levels increased.
Flight day three was dedicated to the first-ever extravehicular activity (EVA) on a commercial spaceflight mission. After extensive preparations, all four crew members donned their EVA suits, which are pressurized with 100% oxygen at 5.1 pounds per square inch (35 kPa). Since the Crew Dragon lacks an airlock, the entire capsule was depressurized during the EVA, exposing all crew members to the vacuum of space, though only two partially exited the spacecraft.

The Apollo astronauts carried about 400 grams of oxygen for the the early missions, with 4 hour EVAs and about 600 for the later mission with 8 hour EVAs. That was their primary oxygen supply, they also carried about 1.8 KG of emergency oxygen that would be blown directly over a period of 30 minutes incase of PLSS failure or suit rupture. That oxygen include allowance for suit leakage.I think that would be human consumption over twenty four hours, in which case the four hour duration oxygen bottle just squirts whiffs into a volume of air to regenerate it.
Though, as I recall, when I looked it up some while back, the supposed requirement was higher.
The equivalent could be a firefighter, without toxic smoke.

Even with a dead hull there would be a substantial amount of Oxygen on board, Oxygen expands about 860 times from liquid form to gaseous form at standard pressure. Nitrogen is similar.I think we have rebreathers.
With an intact hull, but dead air, that would be a question of duration, before a recharge, which probably be some chemical potpourri cachet.
They actually produce a good bit more than one hours oxygen, because the passengers on airplanes won't be rebreathing the oxygen. They make about 250-300 liters of atmospheric oxygen, or about 1/2 to 1/3 of a days oxygen. But since it's not rebreathed it does not last for 8-12 hours, and it is shared between passengers.Oxygen Candles can provide 1 person about ~1 hour of oxygen for consumption.
The chemical reaction is quite exothermic however (600º C).
Their use is typically reserved for environments that have to be sealed (submarines, space stations, etc.).
Scrubbing doesn't need to be chemical based, and IRL chemical cannisters have been phased out for a while. Apollo used Lithium Hydroxide canisters, at a rate of about 2 per day. Skylab switch to regenerative molecular sieves which would capture CO2 for 12 hours and then be renewed by being exposed to vacuum for 12 hours. The Shuttle still used Chemical Absorption, but ISS uses the sieves as will Orion. They do have chemical absorption canisters as an emergency back-up.As I recall, Traveller asphyxiation rules vary.
One variant is oxygen gel, which, if there was a choice, I'd mix with a non toxic substance that would make it non flammable.
At the opposite end is weight, since you could try compressing pure oxygen, and placing in a puncture proof bottle.
Scrubbing should be one and done, since the chemicals would be altered.
However, some advanced form of plant photosynthesis, that speeds up the process, and converts carbon dioxide to breathable oxygen.
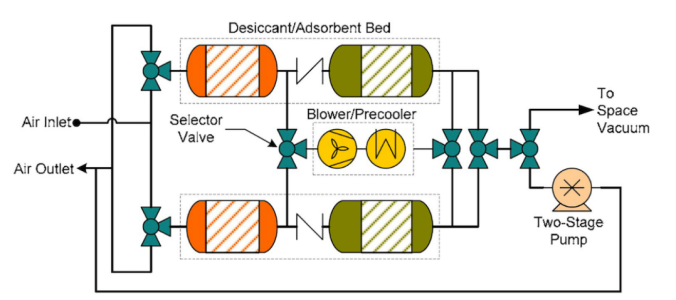
It might be posible to generate sodium chlorate (to make oxygen candles) with the otherwise unused oxygen from the fuel hydrolysis process, given saltwater feedstock and some additional processing equipment.There's more than one way to skin a cat, and considering that breathing would be considered in most cases existential, it's likely that most starships have several ways to regenerate oxygen onboard, or, at least, one backup plan.
Obviously, it would seem that starships while processing unprocessed fuel to processed fuel, from unprocessed water, would use siphon off the byproduct oxygen, into a separate tank under pressure.
If they're using the same procedure for just in time power plants, the oxygen byproduct might almost immediately get mingled with the general onboard air pool.

Given how the body reacts to high CO2 levels, they'll be panicking and in great distress long before they go unconscious, too.CO2 would be a bigger issue, safe levels are mell below 1%. OSHA permits O.5% exposure for 8 hours, and you will see unconscious somewhere above 5%. Which would be 700 liters. About 1.5 times what you exhale in one day. (450-500 liters)
Without scrubbing a person in a one dTon space would exhale enough CO2 to pass out after a day or a day and a half.
SO CO2 scrubbing will define come an issue before Oxygen use does.
